Choosing the right materials can have a major effect on the success and performance of an object. Which material is best to build the frame for a road bike? What material should we use to build a tennis racket? Does it matter what material is used to make an artificial hip? How do I select a material to construct a safety helmet? This topic helps learners understand why a material is selected for a purpose.

|
Created by Mr S Lee
about 8 years ago
|
|

Ionic bonding occurs when the following types of elements bond to form a
compound:
A metal & metal
B metal & non-metal
C non-metal & non-metal
When an ionic compound is formed, the metal atoms:
A gain electrons to form negatively charged ions
B lose electrons to form negatively charged ions
C gain electrons to form positively charged ions
D lose electrons to form positively charged ions
The chlorine atom has the electronic configuration 2,8,7.
The electronic structure of the chloride ion is:
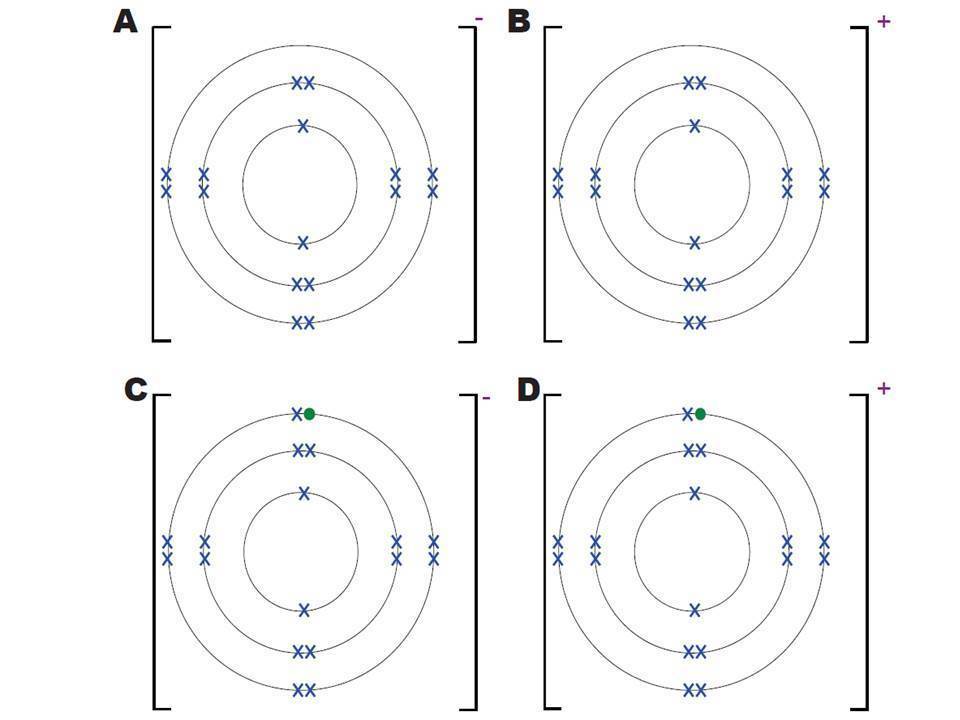
Describe the structure of
sodium chloride (NaCl)
Covalent bonding occurs when the following types of elements bond to
form a compound:
A Metal and a metal
B Metal and a non-metal
C non-metal and non-metal
A water molecule (H2O) consists of
2 hydrogen atoms and 1 oxygen atom.
Draw a dot & cross diagram to show the covalent bonding in a water molecule.
The element carbon comes in different forms or ‘allotropes’. These include:
• graphene
• graphite
• carbon nanotubes
• fullerenes
• diamond
What is an allotrope?
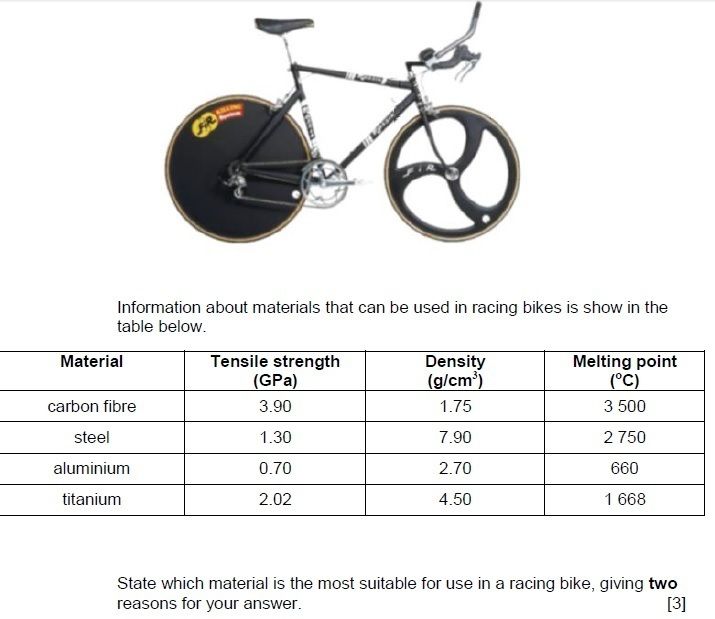
What is an alloy?
Malleability describes the property of a substance that can be:
A pulled into wires
B hammered into sheets
C shattered into pieces
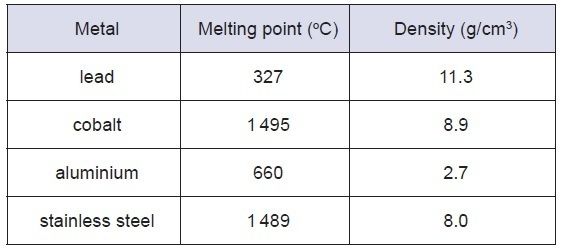
A block of metal has has a mass of
90.0 g and sides of the same length (2 cm).
Identify the metal in the block.
Graphene is an allotrope of carbon. Carbon is a non-metal element.
Graphene sheets are about 200 times stronger than steel and also have a much lower density.
Suggest how these properties make graphene useful for manufacturing car parts.
What are polymers?

Give one example of a polymer and it's uses.
Fill in the gaps using these words: plastic, elastic, do, do not
If a material goes back to its original shape, it has ______________ behaviour. In this case the particles _____________ go back to their original positions.
If a material does not go back to its original shape, it has _______________ behaviour. Now the particles _____________ go back to their original positions.
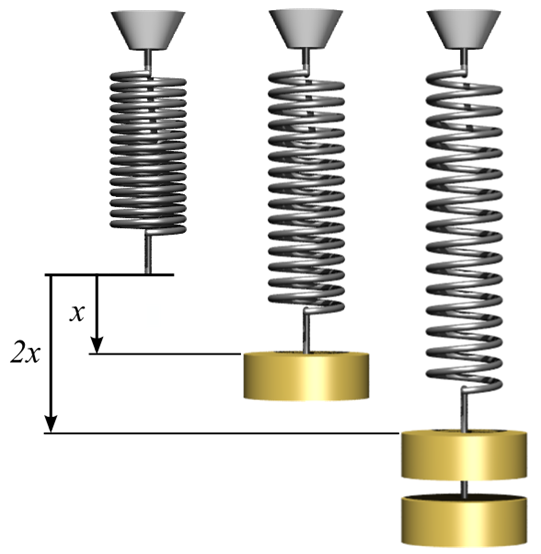
A 20N force stretches a spring by 0.5m.
a) Calculate the spring constant.
b) How much force must used to stretch the spring by 1.5m?

 Hide known cards
Hide known cards
