3 Williams Flashcards on 3prof Williams, created by Magda Sroka on 15/10/2017.
Pinned to
336
1
0
No tags specified
|
|
Created by Magda Sroka
over 8 years ago
|
|
Close
|
|
Created by Magda Sroka
over 8 years ago
|
|

The mechanism of reaction A is known, what do u know about mechanism of reaction B?
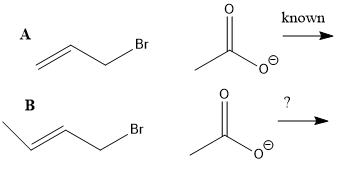
can u say that the reaction proceeds via Sn1?
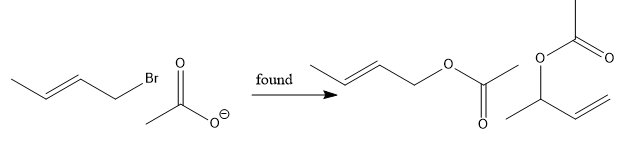
Reaction of CH2=CHCH2Br with acetate gives the same product no matter if the reaction proceeds via Sn1, Sn2 or Sn2’.How to find out which mechanism is taking place? (note what result u would expect in each case and what would u observe in case where more than one mechanism operates
why both of these mechanisms seem reasonable and what test can be done to find out which mechanism takes place?
what tests can u suggest? what outcome u expect from each mech. u propose?(only sketch of and answer is provided here)
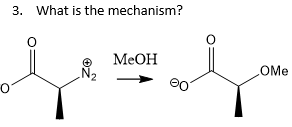
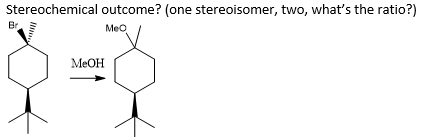
how would u test is a reaction is inter- or intermolecular?
what is e. e. ?
how would u describe the reactions in terms of stereospecificity and stereoselectivity?
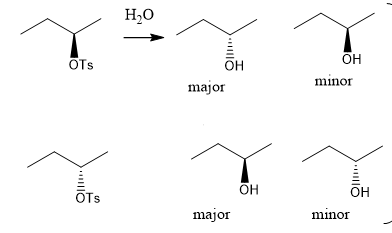
when complete retention of chirality (i.e. a single enantiomer of the product forms) occurs, what can u deduce? (what reaction schemes r consistent with this, which u should exclude?)
what can u deduce about mechanism when there is total loss of chirality? (i.e. racemate of the products forms)
Partial loss of chirality (i.e. a mixture of enantiomers of the products forms).
conditions required for an intermediate
how would u check if the reactions is intra- or inter molecular?
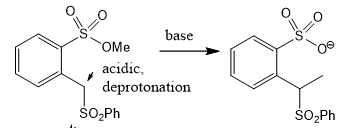
what can u do if u suspect an intermediate?
what does isosbestic point tell u?
can u derive a proof? ;D
what is TS
A. when it is good to use UV to track your reaction?B. how would u find out that u have a mass imbalance in you reaction during UV experiment. C. what would it tell u?
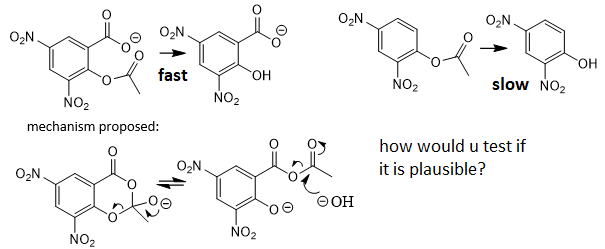
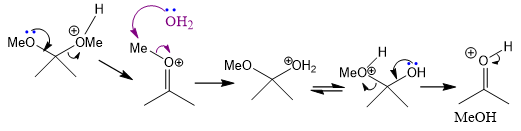
propose mechanism of hydrolysis of 2 consistent with the solvent effect
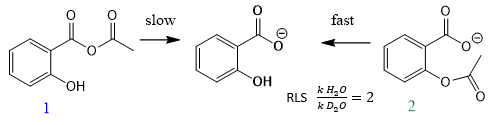
Does the strategy of generating an intermediate separately only hold to stable intermediates? yes? no? can u think of any example?
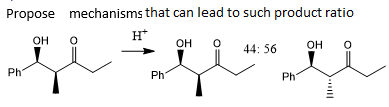
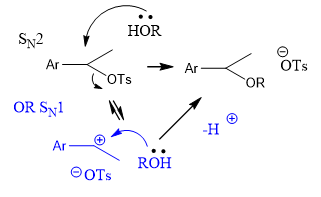
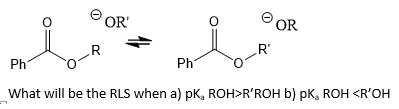
how your u discriminate between these two mechanistic possibilities?
what is Ea in the plot below (from SM to T1/2 or 3?)
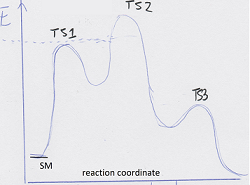
what is the principle of microscopic reversibility?
bearing in mind the principle of microscopic reversibility, what is likely to be the RLS in the above reaction?
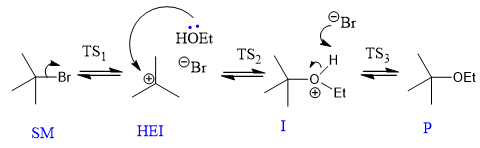
what is Hammond postulate?
what is Curtin-Hammett principle? can u give an example of a reaction when it can be applied? and example where it cannot?
what is Eyring eq,
can u derive Eyring eq?
what is enthalpy of activation related to?
what information do u get form different values of entropy of activation?
what entropy do u expect if the second step is RL, who would u construct ur argument?
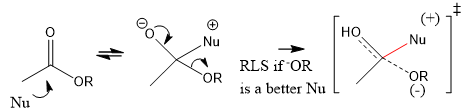
Can species that are taking part in the reaction AFTER RLS appear in the rate law?
B. does solvent appear in the rate law?
can u have a irreversible step BEFORE the RLS?
how many separate mechnisms u have to propose to explain such a rate law?
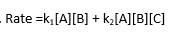
for the scheme above derive rate law for SN1 path knowing that 2nd step is RLS
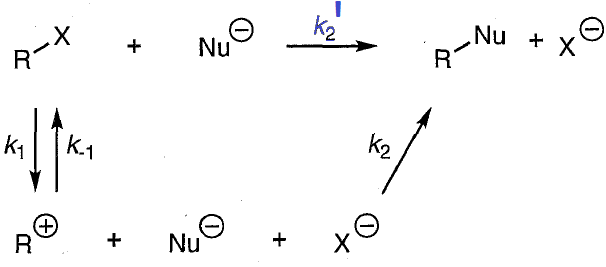
for the scheme above derive a rate law for the SN1 reaction (in general terms, assume that u do not know which step is RLS)

For the reaction below Nu is also a solvent, how would u establish if the mechanism is sn2, sn1 (1step RL or 2nd step RL?)
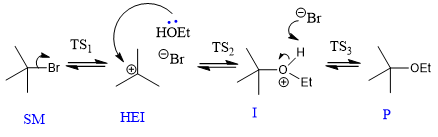
reasons for change in log kobs?
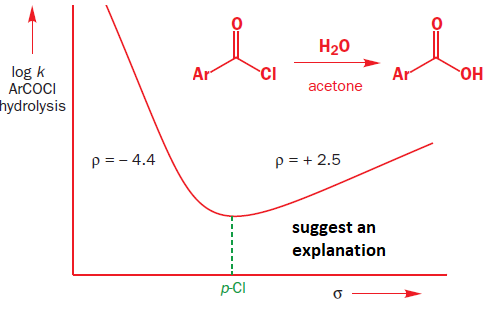
what is strange in this profile and how can u explain in?
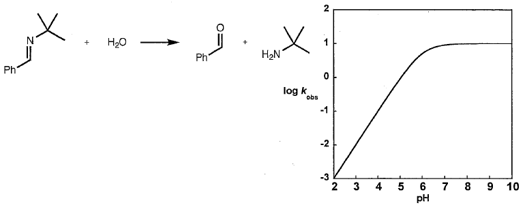
what the magnitude of the primary isotope effect can tell you about the nature of TS?
what is a secondary isotope effect? what magnitude u can expect in different cases?
what isotope effect kH/kD u would expect in E1 reaction?
what do different values of isotope effect tell u?
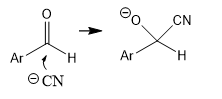
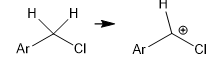
what isotope effect do u expect for the circled H?
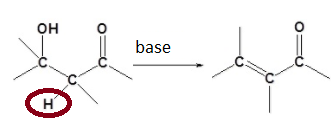
what rho sign and magnitude (very roughly) would u expect for the reaction above?

what rho sign and magnitude (very roughly) would u expect for the reaction above?
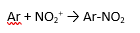
what rho sign and magnitude (very roughly) would u expect for the reaction above?
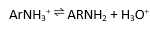
what rho sign and magnitude (very roughly) would u expect for the deprotonation of the above compound?
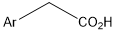
describe steps of constructing a Hammett plot for the above reaction
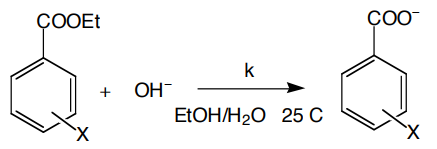
what 2 crucial things u learn form Hammett plots?

what two main information u get from Bronsted plots?
why in the extended Bronsted plots u often get values greater than 1?
kinetic isotope effect?
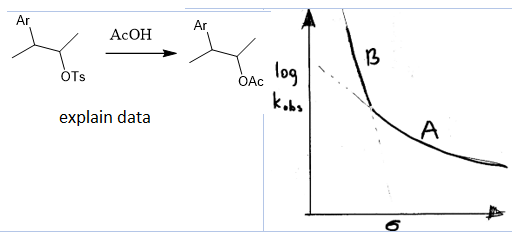
why concave downwards plots tell u about change in the RLS?
kinetic isotope effect?
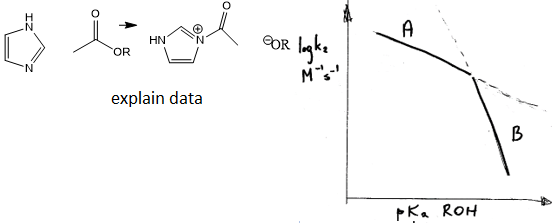
suggests an explanation
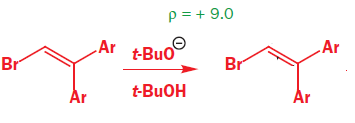
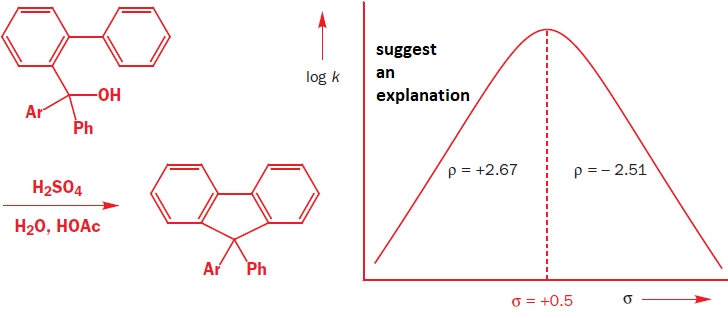
is it likely that removal of a proton form a heteroatom by heteroatom base can be RLS? how about proton removal from carbon?
advantages and disadvantages of general acid/base catalysis

 Hide known cards
Hide known cards