University Organic Chem Flashcards on Structure Determination, created by julia_reast_93 on 17/01/2014.
Pinned to
99
0
0
No tags specified
|
|
Created by julia_reast_93
over 10 years ago
|
|
Close
|
|
Created by julia_reast_93
over 10 years ago
|
|

how do you calculate empirical formula?
How do you obtain the molecular formula?
Alternative method to calculate molecular formula
How do you find the number of double bond equivalents?
1 DBE
2 DBE
4 DBE special case
Beer-Lambert Law
IR
600-1400 cm-1
IR
1600- 1850 cm-1
IR
2100-2300 cm-1
IR
2700-3000 cm-1
IR
3000-3200 cm-1
IR
3200-3450 cm-1
H NMR
δ7-δ8
H NMR
δ5-δ6
H NMR
δ2.5
H NMR
δ9-δ10
H NMR
δ10+
H NMR
~δ1-2
H NMR
~δ2.5
C NMR
δ165-210
C NMR
δ100-150
C NMR
δ65-90
C NMR
δ0-40
C NMR
δ75-95
C NMR
δ110-150
C NMR
δ30-65
H NMR
Withdrawing groups next to the proton
H NMR
Donating groups next to H
If resonance can occur such as a double bond near a carbonyl it is often the H nearest to the opposite end of the carbonyl that is more deshielded rather than the H nearest the withdrawing O because of the resonance sturucture. The same can be said for the Hs near donating groups. See pics
Three factors that affect J (coupling constant)
roofing
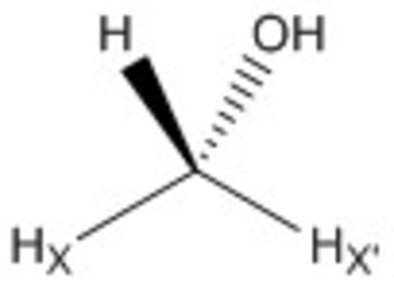
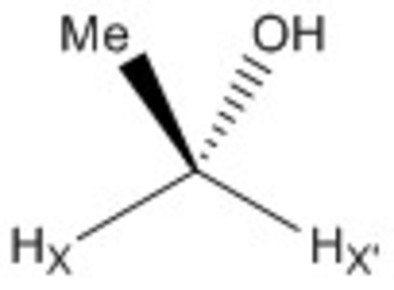
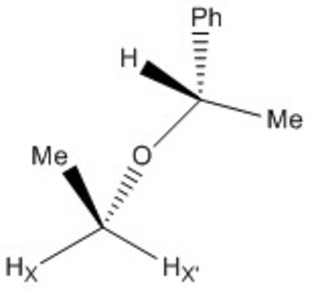
1H NMR exchange
13C NMR
Electronegative substituents
Intensity of signals in 13C NMR
13C-13C coupling
13C-1H coupling
13C-1H off resonance coupling

 Hide known cards
Hide known cards