Flashcards on Unit 5 - transition metals, created by jasmin.sahota on 16/06/2013.
|
|
Created by jasmin.sahota
over 11 years ago
|
|

definition of a transition metal is...
is zinc a transition metal?
is scandium a transition metal?
the physical properties of transition metals from titanium to copper are...
what is the reactivity like in transition metals
for the electronic configuration of transitions metals, all of them have two outer 4s electrons in them except...
generally as we go across the transition metals
describe the electron configuration of chromium
describe the electron configuration of copper
do the electronic configuration with a full 3d and 4s sub level
what are the 4 chemical properties of transition metal?
what does variable oxidation states mean?
if an element has many oxidation states, what does this mean?
what does colour mean as a characteristic of a transition metal?
what does catalysis mean?
where is iron used as a catalyst
where is vanadium oxide used as a catalyst
what is manganese oxide used as a catalysis for?
what does complex formation mean?
when is a complex ion formed
how are the ligands bonded to the transition metal in a complex ion?
[Cu(H20)6]2+ is an example of a
all transition metals can form co-ordinate bonds by
a ligand is
4 examples of ligands are:
the co-ordination number
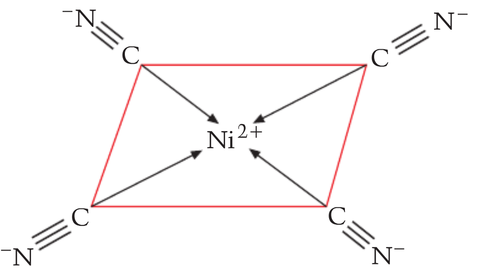
an ion with an octahedral shape has
a tetrahedral shape is
[Co(NH3)6]3+
[CoCl4]2- is an example of
draw [Co(NH3)6]3+
draw [CoCl4]2-
Multidentate ligands are
ethane-1,2-diamine is an example of
draw ethane-1,2-diamine
benzene-1,2-diol is an example of
draw benzene-1,2-diol
ethanedioate ion, (C2O4)2-, is an example of a
draw ethanedioate ion, (C2O4)2-
(EDTA)4- is an example of a
(EDTA)4- is called
draw (EDTA)4-
(EDTA)4- can act as a
chelates are
chelate can be used to
haemoglobin is
haemoglobin is responsible for
haemoglobin consists of an
from the 6 co-ordination number, 4 of them
all ligands are
Cl- ion is a bigger ligand than the ammonia and
the oxidation state of the metal ion =
work out the oxidation state of [CoCl4]2-(aq)
most complexes with four ligands usually have a tetrahedral shape, however
complexes with two cordinate bonds are
cisplatin (Pt(NH3)2Cl2) is square planar
what is cisplatin?
how does cisplatin work?
cisplatin has a isomer called
transplatin looks like
what effect does trans-platin have on cancer?
the downside of cis-platin is...
why are transition metal complexes coloured?
the frequency of the light is =
the frequency is related to the
violet is
red is
the colour of the transition metal complexes depends on
what can we use to find the formula of a transition metal complex?
how does a colorimeter determine the formula of the complex?
the results of the colorimeter are plotted on a graph
iron has two stable oxidation states
Which oxidation state of iron is less stable, Fe2+ or Fe3+?
show the equation and the changing oxidation states when Cl reacts with Fe2+
Cl oxidation number drops from 0 to -1, while Fe2+ goes uo from +2 to +3. what is Cl acting as
Potassium manganate (VII) acts as an oxidixing agent in acidic solution and can oxidise
in the reaction between maganese and iron, the maganeses oxidation state goes from
show the half equation of MnO4- going to Mn2+
and Fe2+ going to Fe3+
and then the overall equation
when maganese is titrated against Fe2+ the endpoint is when
the burette in a titration is filled with
what is in the conical flask of the titration?
why is sulfuric acid added to the reducing agent
draw an example of the titration
Fe2+ can also be oxidised by
the endpoint of potassium dichromate and Fe2+ is when the colour changes
the equation for the reaction of potassium dichromate and Fe2+ is
(Cr2O7)-2
what is oxidation state?
what is the colour of the ion?
(CrO4)2-
what is oxidation state?
what is the colour of the ion?
Cr3+
what is oxidation state?
what is the colour of the ion?
Cr2+
what is oxidation state?
what is the colour of the ion?
summary of the chromate reactions

 Hide known cards
Hide known cards
