Cue cards on Edexcel Unit 2: Nature of Light

|
Created by Niamh Walsh
almost 9 years ago
|
|

Physics Unit 2
Nature of Light

Wave or Particle?
Light shows both wave (it reflects, refracts and diffracts) and particle behaviour (it consists of photons, small packets of energy), so it can be called a wavicle. Einstein discovered the photoelectric effect, which opened up the whole new world of quantum physics
The Photoelectric effect
When UV light increases in intensity, so does the current. However, when light intensity is constant, but the frequency is varied, the current does not increase. No photons are emitted beneath a certain frequency.
Key definitions:
Threshold Frequency: the minimum frequency of light required for photons to be emitted
Work Function (⍉): the minimum energy required to release the photoelectron from the metal
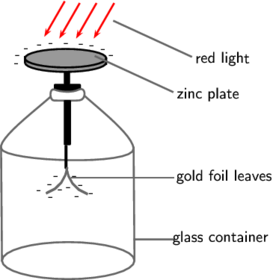
The photoelectric effect supports the particle model for light:
- light is made of photons
- a photon collides with a single electron, giving it the energy needed for it to emit
- electron is emitted immediately
- increased intensity = increased photons
- increased intensity doesn't change max K.E. of electrons
Line spectra: Emission
When light is spread into its seperate colours, it forms a spectrum.
Each line in an emission spectrum arises when an atom drops an energy level. As each material has a unique set of electronic energy levels, each emission spectra is different, allowing us to identify the material

Energy levels show the ground state of atom, which is where it has the least energy. The highest frequency of radiation which could be emitted corresponds to the most energetic photon that could be released.
Solar cells transform light energy into electrical energy
Radiation Flux: the rate at which a beam of light supplies energy to a particular area
F = P/A
F = radiation flux, (Wm^-2)
P = power (W) A = area (m^2)
Laser light is an intense beam, and so it is often used at concerts. The beam is intense because the waves are coherent, and so the photons emitted stimulate other photons, resulting in laser light as the atoms emit photons together

 Hide known cards
Hide known cards
